Call: (800) 382-3176 or Chat with us
Germinal Matrix Hemorrhage and Intraventricular Hemorrhage
Germinal matrix hemorrhage (GMH), a prevalent and concerning issue in preterm infants, has the potential to cause severe complications and long-term effects. Understanding GMH is vital for healthcare professionals, parents, and anyone working with premature infants. In this blog post, we will explore the intricacies of GMH, including risk factors, diagnosis, complications, prevention, management, and legal implications. By the end, you will be well-equipped with the knowledge necessary to comprehend this crucial aspect of neonatal care.
Blog Takeaways
- Germinal matrix hemorrhage is a common and severe intracranial hemorrhage observed in preterm neonates, characterized by fragile blood vessels and rapid angiogenesis.
- Low gestational age, birth weight, and Apgar scores are risk factors for GMH.
- Diagnosis of GMH requires ultrasonography or CT scanning/MRI. It can lead to long-term effects such as neurologic impairment, hydrocephalus & mortality with legal implications in Florida.
Understanding Germinal Matrix Hemorrhage
Germinal matrix hemorrhage, the most common and significant intracranial hemorrhage observed in clinically unstable preterm neonates, can lead to severe neurologic sequelae, including cerebral palsy, mental retardation, and seizures. Unfortunately, premature infants are particularly vulnerable to GMH, with studies showing that 67% of infants born prematurely at 28-32 weeks and 80% of infants born between 23 and 24 weeks of gestation are susceptible to hemorrhages in the germinal matrix, which can lead to germinal matrix intraventricular bleeding into the lateral ventricles, also known as ventricular hemorrhage.
Two primary factors contribute to the heightened vulnerability of blood vessels in the germinal matrix: the fragile nature of the blood vessels and the rapid angiogenesis that occurs in this region. These factors will be closely examined, shedding light on their contribution to GMH risk.
Fragile Blood Vessels
The blood vessels in the germinal matrix have the following characteristics that contribute to their susceptibility to rupture:
- Primitive structure that cannot be categorized as arterioles, veins, or capillaries
- Immature structure and various stages of involution
- Lack of fibronectin
- Fewer pericytes
- Diminished perivascular coverage by glial endfeet
These characteristics are consistent with the regional vulnerability of the blood-brain barrier.
Early diagnosis of GM-IVH is crucial, as it may offer a critical opportunity for the implementation of neuroprotective interventions, potentially leading to improved outcomes. Grasping the fragile nature of these blood vessels empowers healthcare providers to make informed decisions about diagnosing and treating GMH in preterm infants.
Rapid Angiogenesis
Rapid angiogenesis in the subependymal germinal matrix plays a significant role in the vulnerability of blood vessels and the risk of hemorrhage. Vascular endothelial growth factor (VEGF) and angiopoietin-2 induce angiogenesis in the germinal matrix, promoting the sprouting of immature vessels devoid of both basement membrane proteins and pericytes, providing additional evidence for microvascular vulnerability.
The rapid sprouting of immature vessels in the germinal matrix increases the risk of parenchymal hemorrhage due to the fragility of the vasculature in this region. Recognizing rapid angiogenesis’s role in GMH development aids healthcare providers and parents in formulating strategies to prevent and manage this potentially devastating condition.
Risk Factors for Germinal Matrix Hemorrhage
Low gestational age and low birth weight are significant risk factors for germinal matrix hemorrhage. Infants with GMH tend to have significantly lower gestational ages and birth weights, and these factors are considered perinatal risk factors for GMH. Additionally, a low Apgar score, a vital assessment of a newborn’s overall health, at 1 and 5 minutes after birth is correlated with an increased risk of GMH.
Gestational Age and Birth Weight
There is a correlation between low gestational age and germinal matrix hemorrhage. Infants with lower gestational ages are at an increased risk of developing GMH. Low birth weight is also a risk factor for GMH, and delivery practices and indications, as well as genetic factors, can contribute to the risk. Very low birth weight infants who carry prothrombotic mutations have been found to be at a heightened risk of developing GMH.
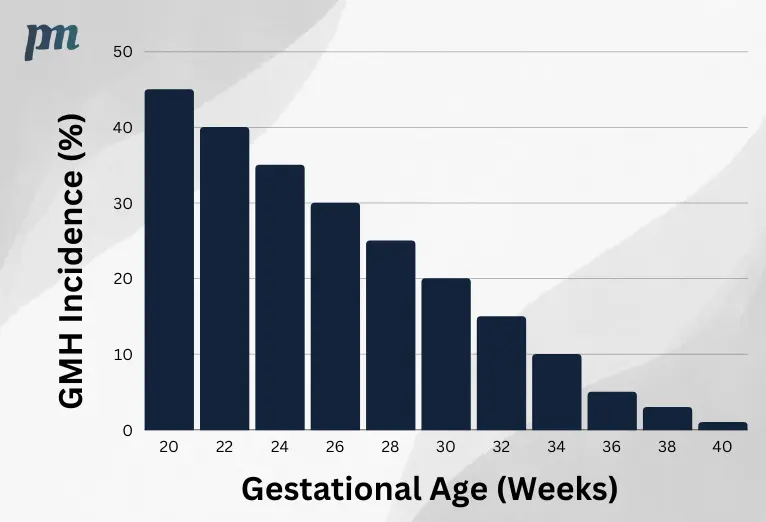
Statistical data on GMH in preterm infants reveals that the overall incidence of GMH in preterm infants is approximately 20 to 25% in infants with a low birth weight (less than 1500 g). The severity of GMH is inversely proportional to gestational age and birth weight, meaning that extremely preterm infants, as well as premature infants with low gestational age, low birth weight, and other risk factors, are more likely to experience severe GMH.
Apgar Scores
The Apgar score is a rapid evaluation of a newborn’s overall health and wellbeing. It assesses five factors:
- Heart rate
- Respiratory effort
- Muscle tone
- Reflex irritability
- Color
The score ranges from 0 to 10, with a higher score indicating better health. A low Apgar score can indicate an increased risk of GMH in newborns.
A low Apgar score at 1 and 5 minutes after birth is associated with an increased risk of germinal matrix hemorrhage (GMH), a type of bleeding in the brain that can occur in premature infants. Thus, healthcare providers should be attentive to low Apgar scores as a potential indicator of risk for GMH.
Diagnosing Germinal Matrix Hemorrhage
Ultrasonography is the most recommended approach for diagnosing germinal matrix hemorrhage in newborns. It is crucial to classify and use grading intracranial hemorrhage techniques, such as GM-IVH, to determine severity and potential outcomes for affected infants.
Early detection of GMH is paramount as it could trigger neuroprotective interventions and potentially enhance outcomes.
Ultrasonography
Ultrasonography, also referred to as ultrasound or sonography, is a noninvasive imaging test that utilizes high-frequency sound waves to generate images of internal organs and tissues. It enables healthcare providers to visualize inside the body without the necessity for surgery.
Ultrasonography is the preferred method for diagnosing GMH due to its noninvasive nature, the ability to perform it at the bedside, and the capacity for repeated assessments.
CT Scanning and MRI
Although CT scanning and MRI offer higher sensitivity than ultrasonography, they require moving the infant from the nursery, making them less practical for diagnosis. CT scanning is capable of demonstrating regions of high attenuation indicative of hemorrhage in the brain, while MRI is particularly beneficial in evaluating infants with germinal matrix hemorrhage.
Complications and Long-Term Effects
Comprehending the potential complications and long-term effects of germinal matrix hemorrhage is crucial. For grades III and IV GMH, there is a high incidence of neurologic impairment, hydrocephalus, and mortality. In contrast, grade I and II hemorrhages have a more favorable outlook.
Potential long-term effects of GMH like cerebral palsy and hydrocephalus necessitate vigilant monitoring and follow-up care for affected infants.
Cerebral Palsy
Cerebral palsy is a neurological disorder caused by germinal matrix hemorrhage in newborns, which can affect cerebral blood flow, and is characterized by impaired movement, coordination, and posture, as well as other impairments such as cognitive deficits, speech and language impairments, hearing and visual deficits. The primary indications of cerebral palsy in children resulting from GMH encompass:
- Clinical neurological decline
- Respiratory difficulty
- Apnea
- Protuberant fontanelle
- Convulsions
- Hypoactivity
- Developmental lags
Survivors of GMH and intraventricular hemorrhage (IVH) in cerebral palsy may experience neurodevelopmental impairments, such as motor delays and impairments, cognitive deficits, speech and language impairments, hearing and visual deficits. Germinal matrix hemorrhage can cause various types of cerebral palsy, such as spastic hemiparesis, spastic diplegia, and spastic quadriplegia.
Hydrocephalus
Hydrocephalus is a condition characterized by the accumulation of cerebrospinal fluid in the brain, resulting in increased pressure and enlargement of the lateral ventricle. GMH can lead to the development of hydrocephalus due to disruption of normal cerebrospinal fluid circulation and absorption.
Potential complications associated with hydrocephalus stemming from GMH include:
- Posthemorrhagic hydrocephalus
- Cerebral palsy
- Epilepsy
- Severe neurodevelopmental abnormalities
- Increased mortality.
In order to diagnose and manage hydrocephalus in newborns with GMH, healthcare providers may utilize the following techniques and interventions:
- Imaging techniques such as cranial ultrasound (CUS) or magnetic resonance imaging (MRI) to assess the condition
- Medical interventions to monitor and treat symptoms, control intracranial pressure, and provide supportive care
- Surgical interventions like ventriculoperitoneal shunting in certain cases to manage hydrocephalus
These approaches are used to ensure proper diagnosis, treatment, and management of hydrocephalus in newborns with GMH.
Prevention and Management Strategies
Antenatal care and prenatal interventions may help reduce the risk of germinal matrix hemorrhage in newborns. It’s crucial to manage GMH and mitigate long-term complications through postnatal monitoring and treatment.
Preventing preterm birth stands as the most effective strategy in curbing the incidence of GM-IVH.
Antenatal Care and Prenatal Interventions
The World Health Organization is a strong advocate for the use of corticosteroids during pregnancy between 24 and 34 weeks. This is especially important when premature birth is imminent. Ethamsylate has been shown to significantly reduce the risk of Grade III and IV intraventricular hemorrhage (IVH) in preterm infants. Studies conducted on animals have indicated that prenatal corticosteroids stabilize the GM vasculature by suppressing vascular endothelial growth factor and increasing transforming growth factor- (TGF-) levels.
This leads to angiogenesis inhibition, trimming of neovasculature, and increased pericyte coverage, thus reducing the risk of hemorrhage.
Postnatal Monitoring and Treatment
The recommended postnatal monitoring procedures for germinal matrix hemorrhage involve:
- Screening by ultrasound or brain imaging in the initial 4 to 7 days after birth
- Re-screening between 4 to 6 weeks of age
- Close neuroimaging for detection and follow-up of any complications
- MRI evaluation for infants with post-hemorrhagic ventricular dilatation and periventricular hemorrhagic infarction.
At present, there is no medical treatment for germinal matrix hemorrhage in newborns. The emphasis is on providing supportive care and addressing any symptoms that may appear.
Legal Aspects of Germinal Matrix Hemorrhage
The legal implications of germinal matrix hemorrhage may encompass medical malpractice claims if the healthcare provider does not adhere to the necessary standard of care, thus causing harm to the patient. In accordance with Florida law, medical malpractice is a claim resulting from the lack of or an improper delivery of medical care. In the event of medical negligence leading to GMH in Florida, medical malpractice claims can be filed.
Medical Malpractice in Florida
According to Florida law, medical malpractice is a healthcare professional’s improper, unskilled, or negligent treatment of a patient. Percy Martinez Law offers dedicated service and expertise in civil trial law and medical malpractice injuries when addressing medical malpractice cases in Florida. They provide assistance to clients throughout the process and strive to achieve successful results in a timely manner.
Florida Statutes and Laws
Medical malpractice is defined by Florida law as any legal action for the purpose of obtaining damages for death or personal injury caused by the negligence of a healthcare provider. The statute of limitations for filing a medical malpractice lawsuit in Florida is typically two years from the date of the incident or discovery of the injury.
Blog Summary
Understanding germinal matrix hemorrhage, its risk factors, diagnosis, complications, and legal implications is crucial for healthcare providers, parents, and caregivers of preterm infants. By being well-informed about GMH, we can help ensure that these vulnerable infants receive the best possible care and support to minimize the risk of severe complications and long-term effects. Education, prevention, and management strategies are key to making a difference in the lives of these infants and their families.
Frequently Asked Questions
What is the germinal matrix?
The germinal matrix (GM) is a highly cellular and vascularized region in the developing brain located underneath the lateral ventricles. It is active between 8-36 weeks gestation and produces both neurons and glial cells. GMH, or hemorrhage of this area, is a devastating neurological disease in premature infants.
Does germinal matrix go away?
The germinal matrix is a fetal structure present from 7-8 weeks of gestation, which persists into the beginning of the third trimester before gradually disappearing by 35-36 weeks. By 36-37 weeks gestation, the germinal matrix has essentially vanished.
What is a germinal matrix hemorrhage?
Germinal matrix hemorrhage is a type of bleeding that occurs in the subependymal germinal matrix, which is part of the brain’s lateral ventricles. It typically occurs due to perinatal asphyxia in preterm neonates and is indicated by weak walls in the blood vessels of the germinal matrix that can rupture under significant stress after birth. Diagnosis is typically performed via ultrasound.
What is the prognosis for germinal matrix hemorrhage?
The prognosis for germinal matrix hemorrhage depends on the extent of hemorrhage and the presence of hydrocephalus, with Grade I and II hemorrhages having a good prognosis, and mortality rates of 3.1% (grade I) and 7.8% (grade II). However, higher grade hemorrhages have higher mortality rates, up to 36.1% for Grade IV.
What are the primary risk factors for germinal matrix hemorrhage?
Low gestational age and low birth weight are the primary risk factors for germinal matrix hemorrhage.